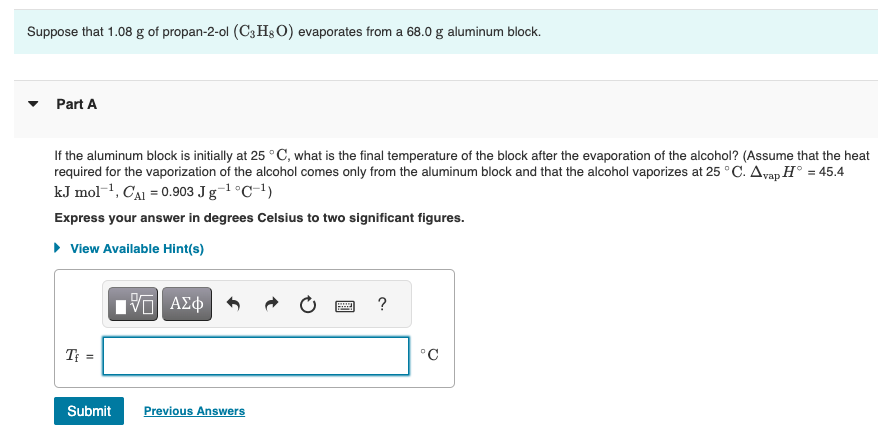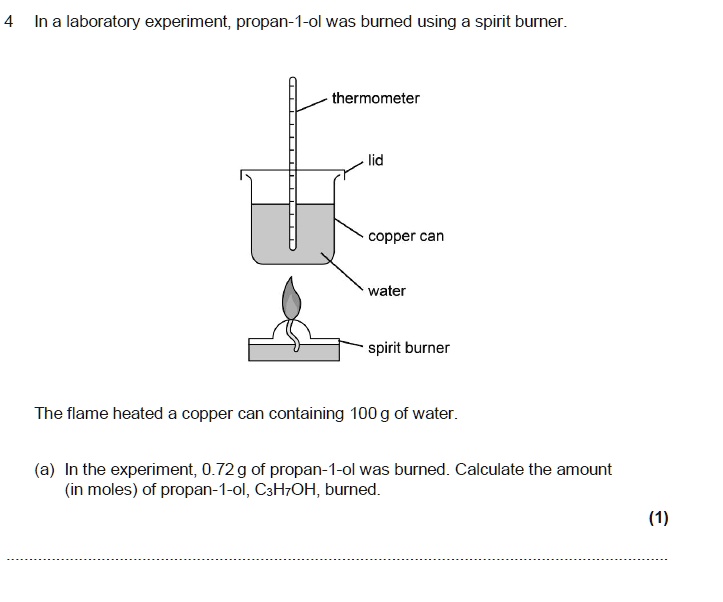
SOLVED: In a laboratory experiment propan-I-ol was burned using a spirit burner. thermometer copper can water spirit burner The flame heated a copper can containing 100 g of water In the experiment;
![3,9-dimethyl-7-[(4-methylphenyl)methyl]-1-(propan -2-yl)-1H,4H,6H,7H,8H,9H-[1,2,4]triazino[4,3-g]purine-6,8-dione | S6705125 | smolecule 3,9-dimethyl-7-[(4-methylphenyl)methyl]-1-(propan -2-yl)-1H,4H,6H,7H,8H,9H-[1,2,4]triazino[4,3-g]purine-6,8-dione | S6705125 | smolecule](https://www.smolecule.com/img/structure/S6705125.png)
3,9-dimethyl-7-[(4-methylphenyl)methyl]-1-(propan -2-yl)-1H,4H,6H,7H,8H,9H-[1,2,4]triazino[4,3-g]purine-6,8-dione | S6705125 | smolecule

G E m against x 1 values for the propan-1-ol(1) + MTBE(2) system: K,... | Download Scientific Diagram
![propan-2-yl 2-chloro-5-[[2-methyl-1-oxo-1-(6H-pyrazolo[3,4-g ][1,3]benzothiazol-2-ylamino)propan-2-yl]amino]benzoate | C22H22ClN5O3S - PubChem propan-2-yl 2-chloro-5-[[2-methyl-1-oxo-1-(6H-pyrazolo[3,4-g ][1,3]benzothiazol-2-ylamino)propan-2-yl]amino]benzoate | C22H22ClN5O3S - PubChem](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=57951425&t=l)




![Buy 4-[1-(propan-2-yl)-1H-benzimidazol-2-yl]butanoic acid (bt-1479884)| THE BioTek Buy 4-[1-(propan-2-yl)-1H-benzimidazol-2-yl]butanoic acid (bt-1479884)| THE BioTek](https://www.thebiotek.com/images/structure/BT-1479884.png)



![GAC82457 | 1256824-57-3 | 3-Propan -2-yl-2H-pyrazolo[3,4-b]pyridine-5-carbaldehyde GAC82457 | 1256824-57-3 | 3-Propan -2-yl-2H-pyrazolo[3,4-b]pyridine-5-carbaldehyde](https://www.biosynth.com/storage/ChemAxonStructureImages/GA/C8/GAC82457_structure.png)

![Buy 3-[4-(Propan-2-yloxy)phenyl]propan-1-amine hydrochloride - 1193388-53-2 | Benchchem Buy 3-[4-(Propan-2-yloxy)phenyl]propan-1-amine hydrochloride - 1193388-53-2 | Benchchem](https://www.benchchem.com/img/structure/B1454891.png)