
SOLVED: Why is nitrite ion used in the oxidation of iodide ion whereas permanganate is used for bromide ion

Permanganate ion reacts with bromide ion in basic medium to give manganese dioxide and bromate i... - YouTube

BEBERAPA OKSIDATOR DALAM LABORATORIUM (ION PERMANGANANAT, ION KROMAT DAN ION KROMAT) | chemistry for peace not for war

Assigning Formal Charges to Each Atom in a Dot Structure Practice | Chemistry Practice Problems | Study.com

SOLVED: (i) In the chromium half equation, the dichromate ion is converted to the chromium (III) ion Cr2O72-(aq) Cr3+(aq) Determine the oxidation number of chromium in each species and hence find the
![🔴[REDOKS]🔴Ion mangan(II) dan ion permanganat dapat bereaksi membentuk MnO₂ dalam larutan.MnO₄⁻ + Mn² - YouTube 🔴[REDOKS]🔴Ion mangan(II) dan ion permanganat dapat bereaksi membentuk MnO₂ dalam larutan.MnO₄⁻ + Mn² - YouTube](https://i.ytimg.com/vi/AGc4mhGxl9U/hqdefault.jpg)
🔴[REDOKS]🔴Ion mangan(II) dan ion permanganat dapat bereaksi membentuk MnO₂ dalam larutan.MnO₄⁻ + Mn² - YouTube
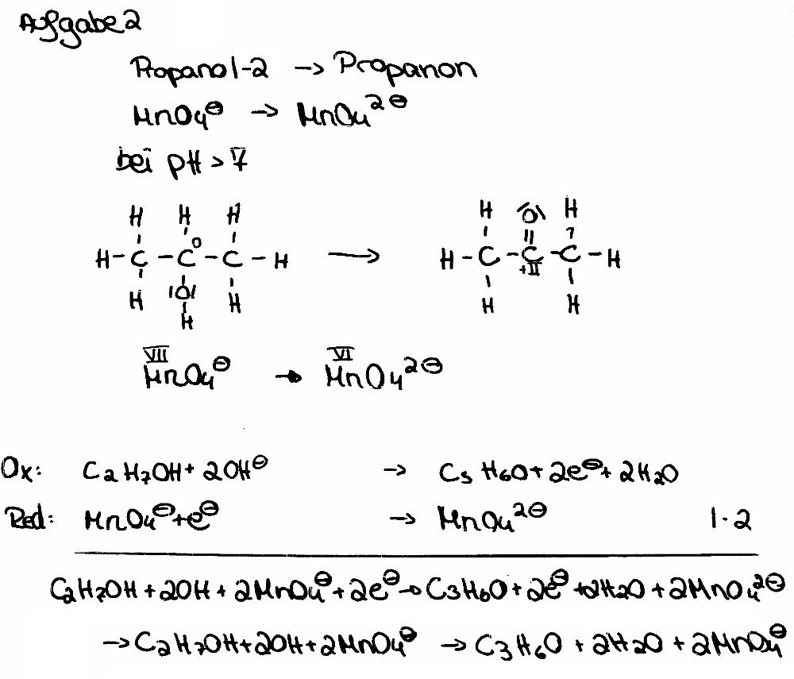










![Kaliumpermanganat • einfach erklärt: Anwendung · [mit Video] Kaliumpermanganat • einfach erklärt: Anwendung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/003/241/af42323d2fc4b132245880626ed02f24b62c8285/Thumbnail_Kaliumpermanganat.png?1665554570)
