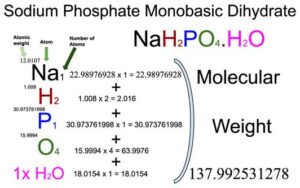
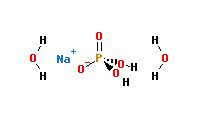
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%20500G.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs
SODIUM DIHYDROGEN PHOSPHATE STARTING FROM SODIUM CHLORIDE AND ORTHOPHOSPHORIC ACID VIA CATION RESIN EXCHANGE Doan Pham Minh, A

Effect of different concentrations of NaH2PO4·2H2O on the cell number... | Download Scientific Diagram
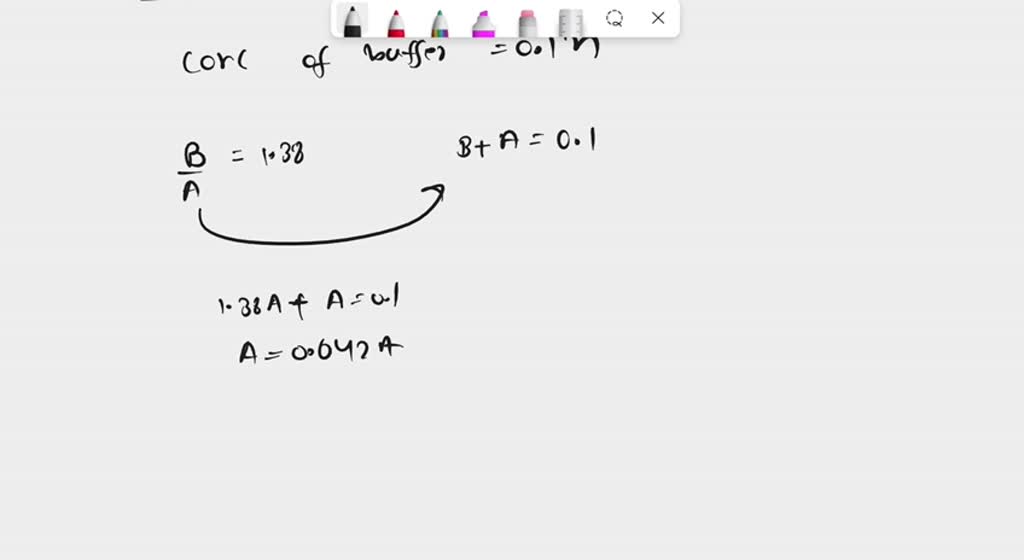
SOLVED: 1. How many g of Na2HPO4 and NaH2PO4 2H2O would you need to prepare 1L of 0.1M sodium phosphate buffer pH 7.0? (Hint= use the Henderson-Hasselbalch equation) Express your answer to

Jual nah2po4 h20 sodium dihydrogen phosphate monohydrate merck 1.06346 - Kota Surabaya - mutiara bersaudara | Tokopedia
Lịch sử giá Hóa chất sodium phosphate monobasic dihydrate nah2po4 cas 13472-35-0 nah2po4.2h2o xylong lọ 500g cập nhật 2/2023 - BeeCost
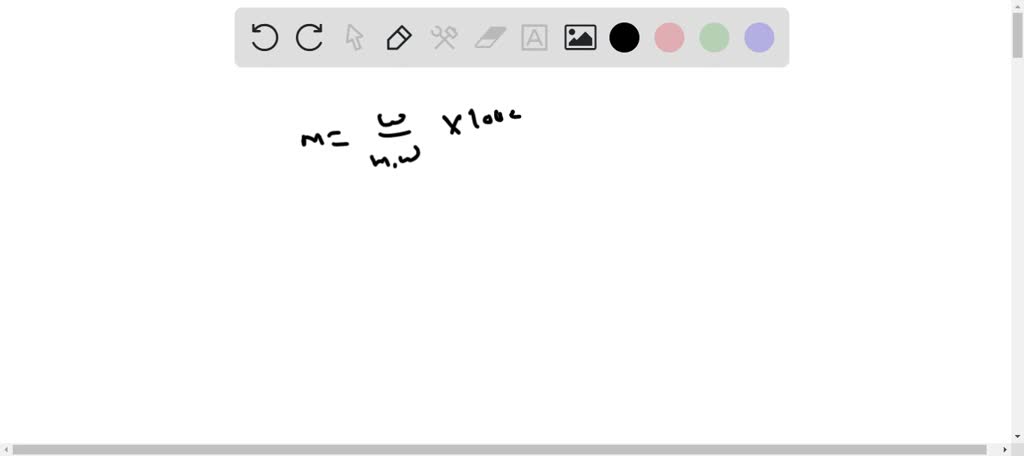










![PART 1: DATA [4 POINTS] Mass of NaH2PO4+H2O = 0.65 g | Chegg.com PART 1: DATA [4 POINTS] Mass of NaH2PO4+H2O = 0.65 g | Chegg.com](https://media.cheggcdn.com/media/1ae/1aecabab-9b80-48fe-99b2-8b304c594dc1/php5IPXYg)
