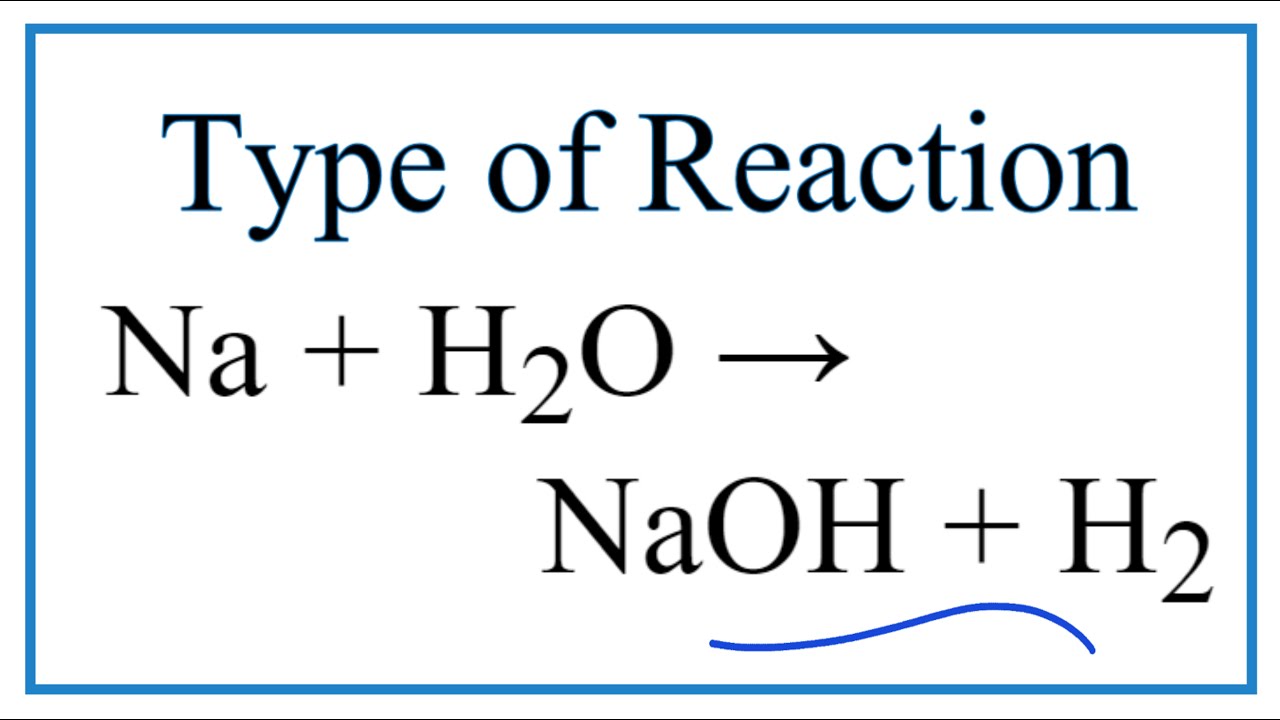
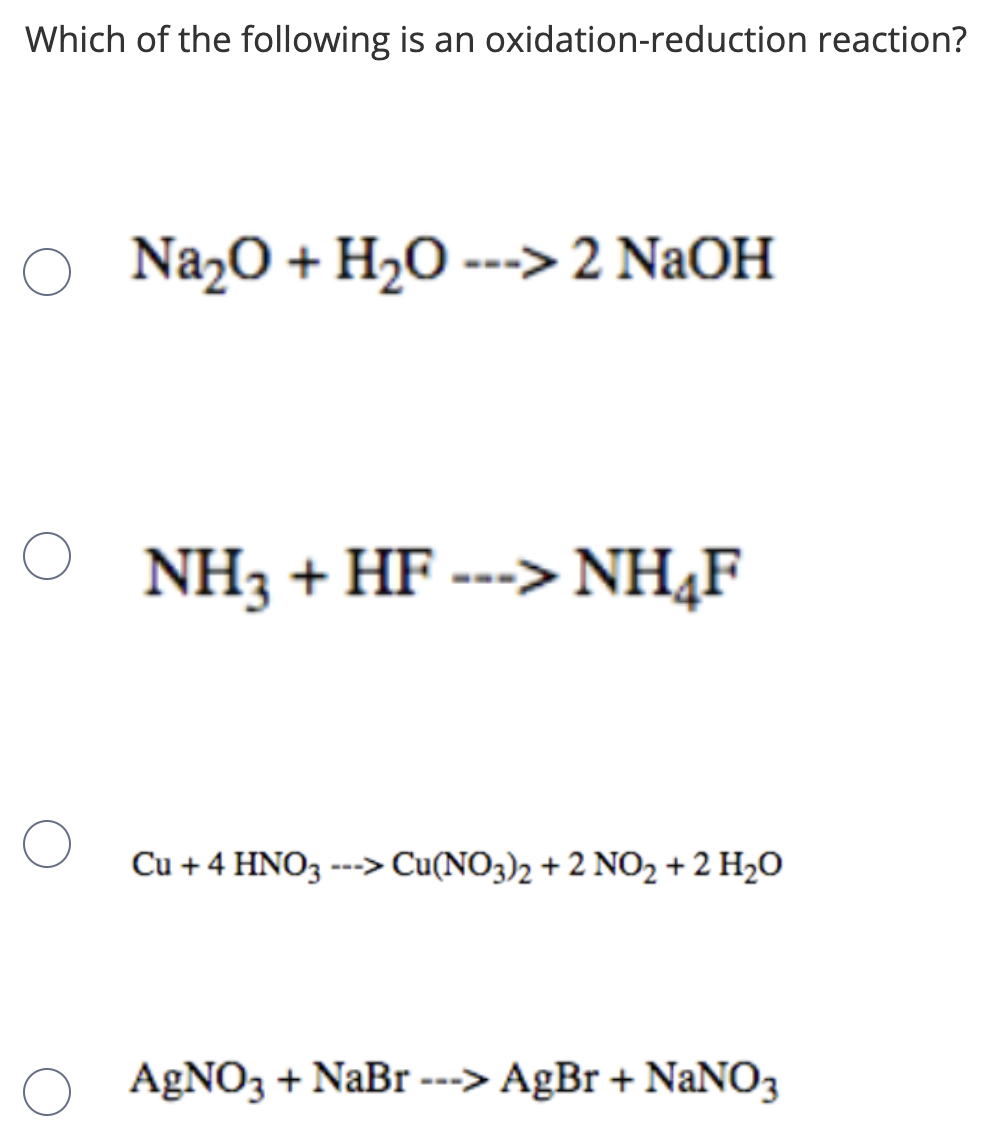Detecting an alkali. Pipette that has been used to add sodium oxide (Na2O) to water (H2O) containing universal indicator. Universal indicator is used Stock Photo - Alamy

Complete the following equation: Na2O2 + 2H2O → (W) + H2O2 2KO2 + 2H2O→ (X) + (Y) + O2 Na2O + CO2→ (Z)
![Effect of Oxygen on Ammonothermal Synthesis: Example of Na2[Zn(NH2)4] ⋅ (NH3)x and Na2[Zn(NH2)4] ⋅ (H2O)x - Kunkel - 2021 - European Journal of Inorganic Chemistry - Wiley Online Library Effect of Oxygen on Ammonothermal Synthesis: Example of Na2[Zn(NH2)4] ⋅ (NH3)x and Na2[Zn(NH2)4] ⋅ (H2O)x - Kunkel - 2021 - European Journal of Inorganic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/6d0a2979-5094-445c-a1c5-5dbc03a1765e/ejic202100721-toc-0001-m.jpg)
Effect of Oxygen on Ammonothermal Synthesis: Example of Na2[Zn(NH2)4] ⋅ (NH3)x and Na2[Zn(NH2)4] ⋅ (H2O)x - Kunkel - 2021 - European Journal of Inorganic Chemistry - Wiley Online Library

EDTA Disodium salt solution for 1000 ml volumetric solution c(Na2-EDTA X 2 H2O) 0.1 mol/l (0.2N) - Th. Geyer

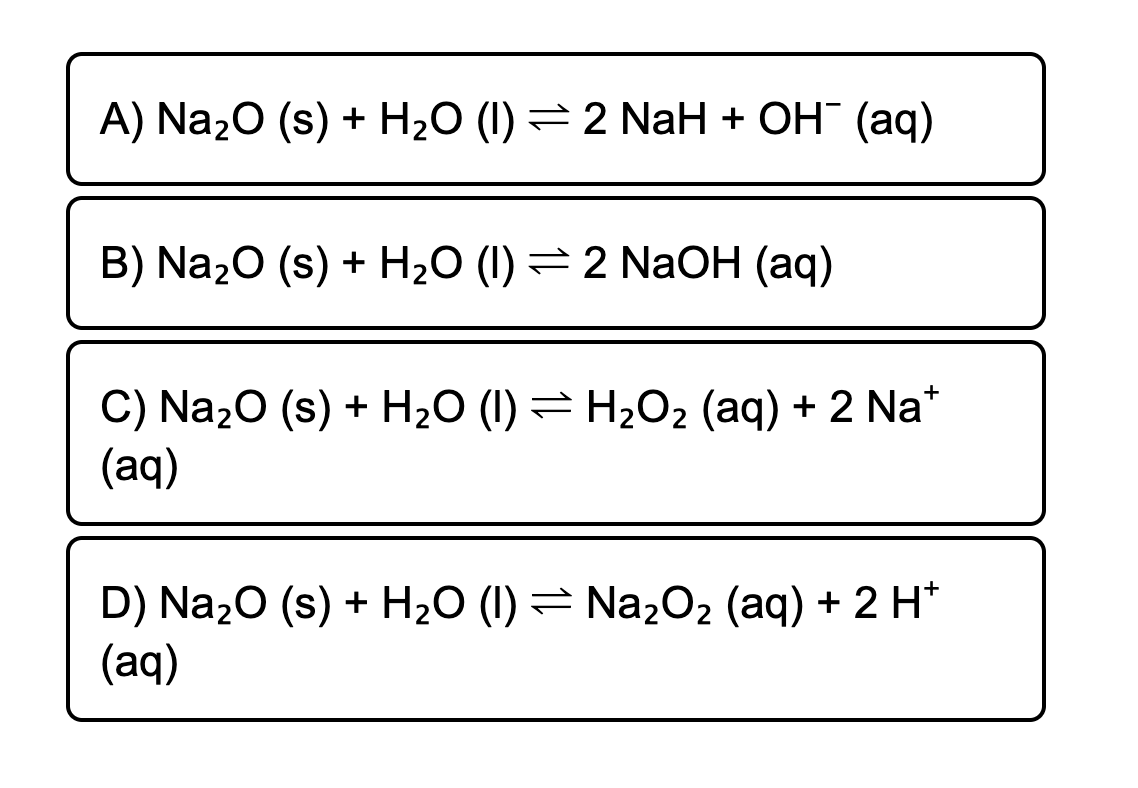
Закончите уравнение и определите тип химической реакции •Na2O + H2O •Na2SiO3 + H2SO4 •Al + - Школьные Знания.com

Comparison of the Coordination of B12F122-, B12Cl122-, and B12H122- to Na+ in the Solid State: Crystal Structures and Thermal Behavior of Na2(B12F12), Na2(H2O)4(B12F12), Na2(B12Cl12), and Na2(H2O)6(B12Cl12). | Semantic Scholar

Q Complete the following equations : (a) Na2 O + H2O → - Chemistry - The s-Block Elements - 12343327 | Meritnation.com
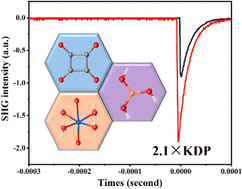
Na2(C4O4)(H3BO3)(H2O)4·H3BO3: the first borate-squarate with strong SHG response obtained by superior synergy - Chemical Communications (RSC Publishing)