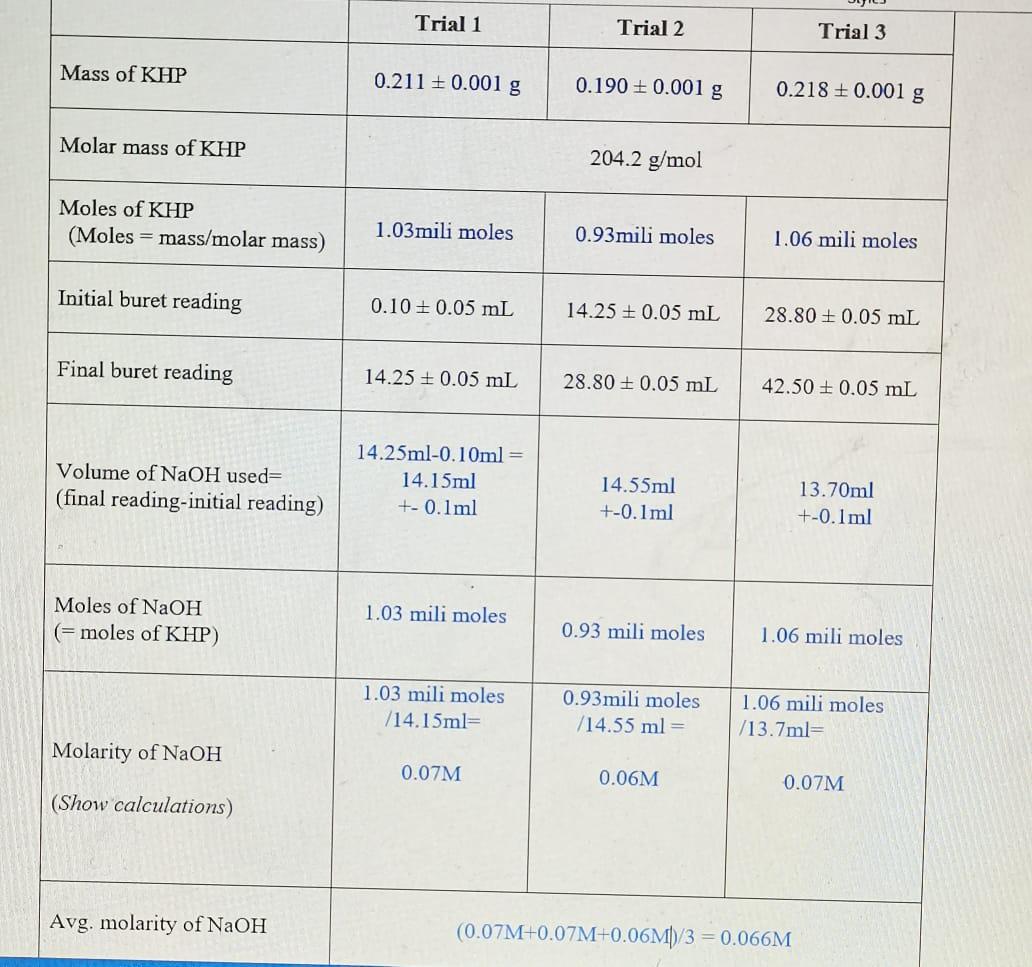
0.1 mili moles of CdSO(4) are present in 10ml acid solution of 0.08 N HCI. Now H(2)S is passed to precipitate all the Cd^(2+) ions. Find the pH of solution after filtering
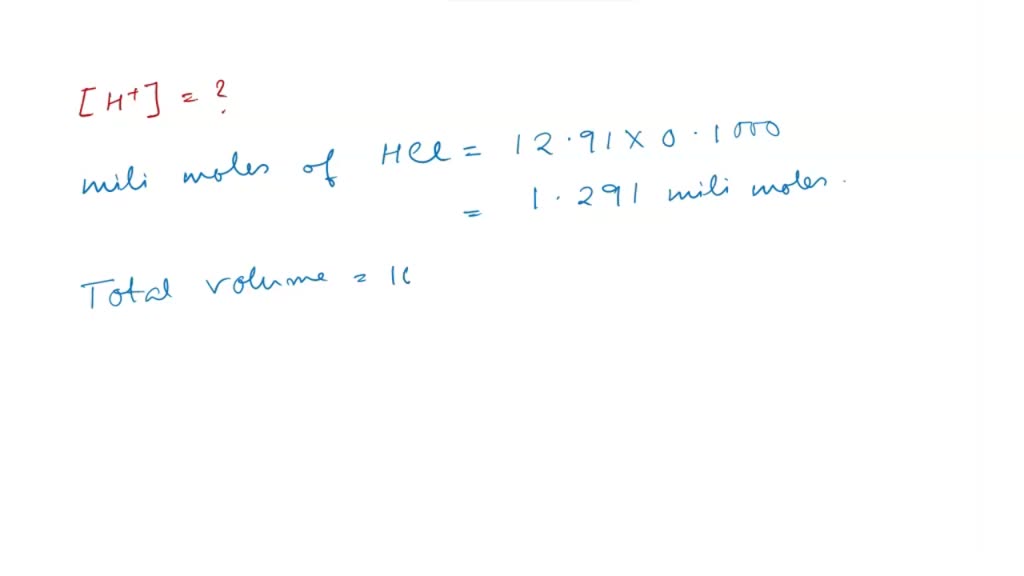
SOLVED: 1. A student mixed the following reactants, using the general procedure for the experiment: 10.24 mL of 0.1000M KI, 24.04 mL of 0.001000 M Na2SO3, 13.82 mL of 0.04000M KBrO3, and

JUKI MOL 254 katlama mili rotor G5061 254 000 silindir endüstriyel dikiş makinesi yedek parçası| | - AliExpress

SOLVED: The percent by mass of phenol (MM = 94.11 glmol) in an aqueous solution is 10.9%. What is the molality of the phenol solution? 0 of 1 point earned 3 attempts