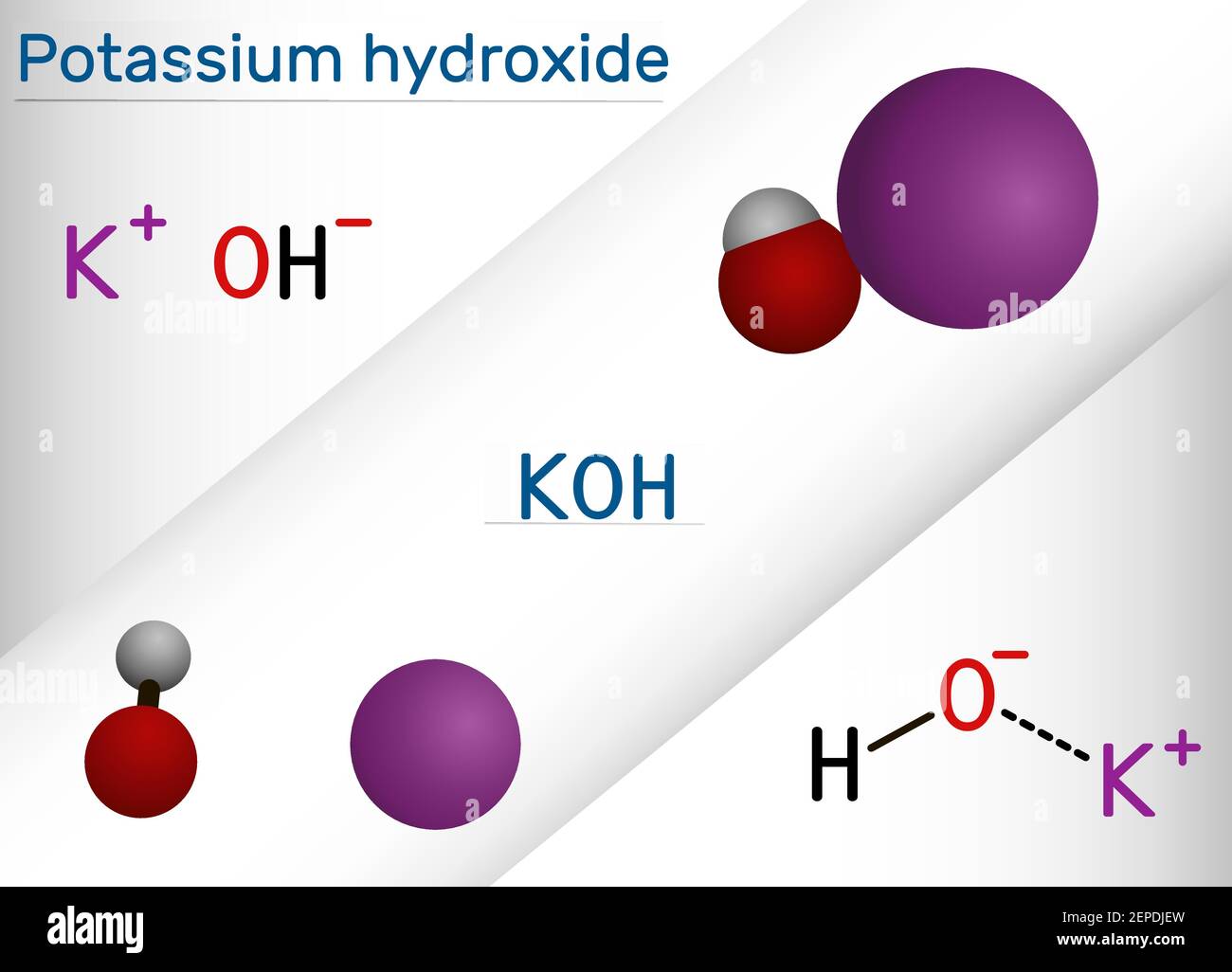
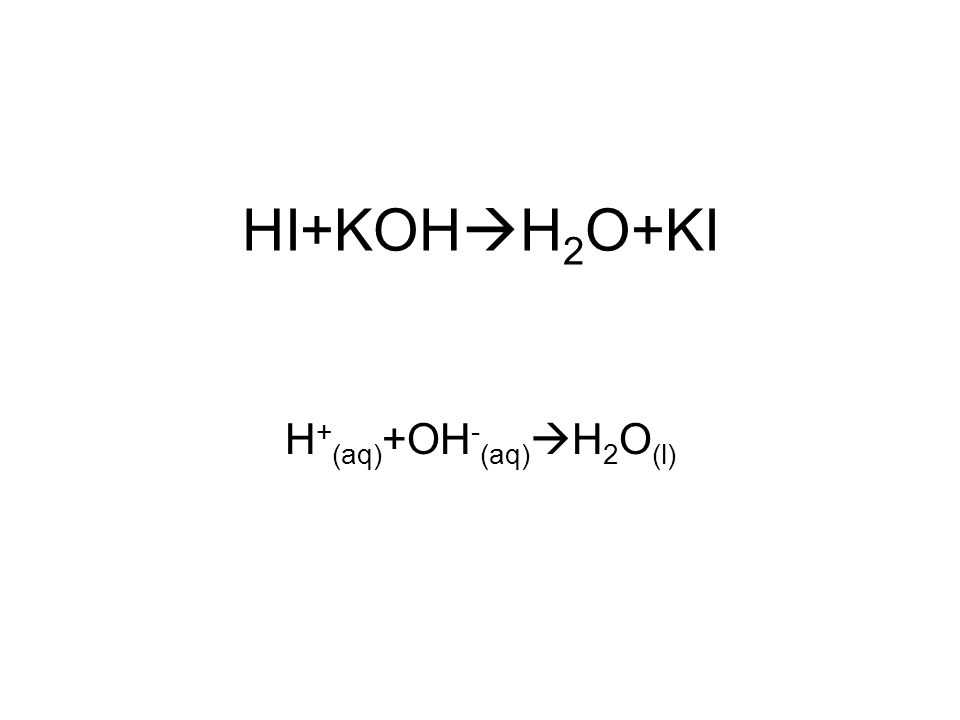
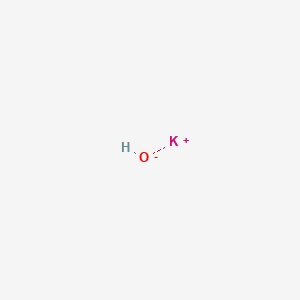SOLVED: Potassium hydroxide dissociates in water to produce hydroxide ions KOH(s) +HzO() Kt(aq) + OH (aq) Given the information above, how is potassium hydroxide categorized? Both a Bronsted-Lowry base ad an Arrhenius

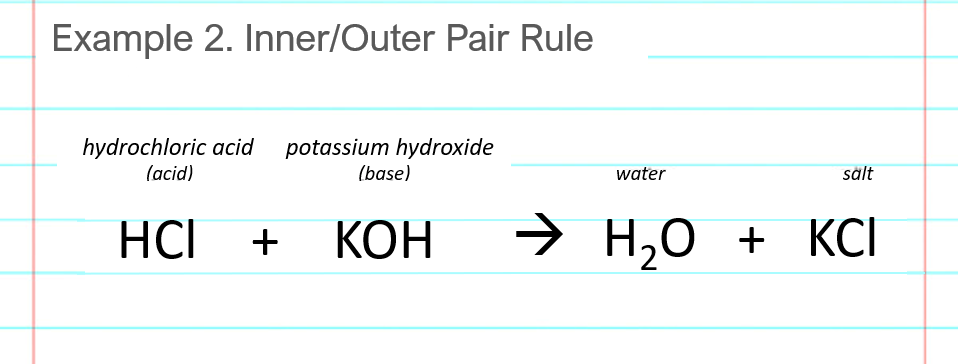
SOLVED: List the conjugate acid or conjugate base for each chemical. a. The acid HF b. The base KOH c. The base NH3 d. The acid HNO3 e. The acid HCOOH f.
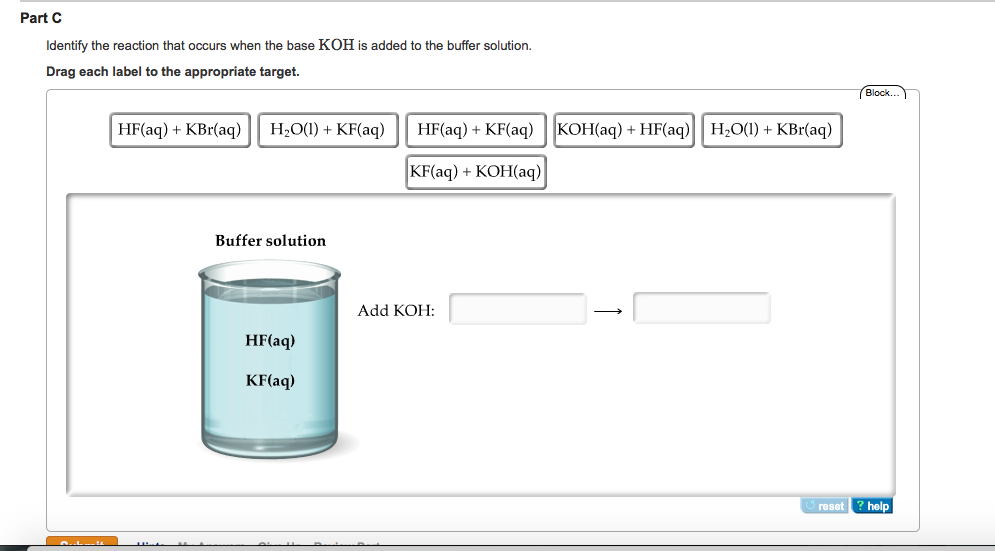
![Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/buffer_solution613505384566154416.jpg)
Identify the reaction that occurs when the base KOH is added to the buffer solution. [{Image src='buffer_solution613505384566154416.jpg' alt='buffer solution' caption=''}] | Homework.Study.com