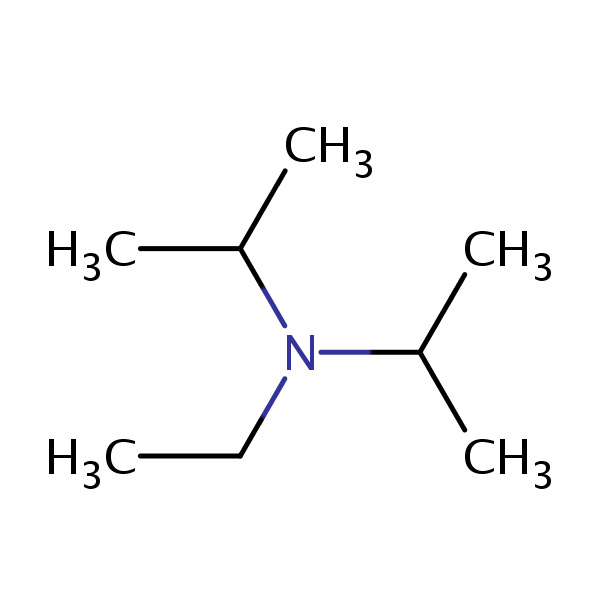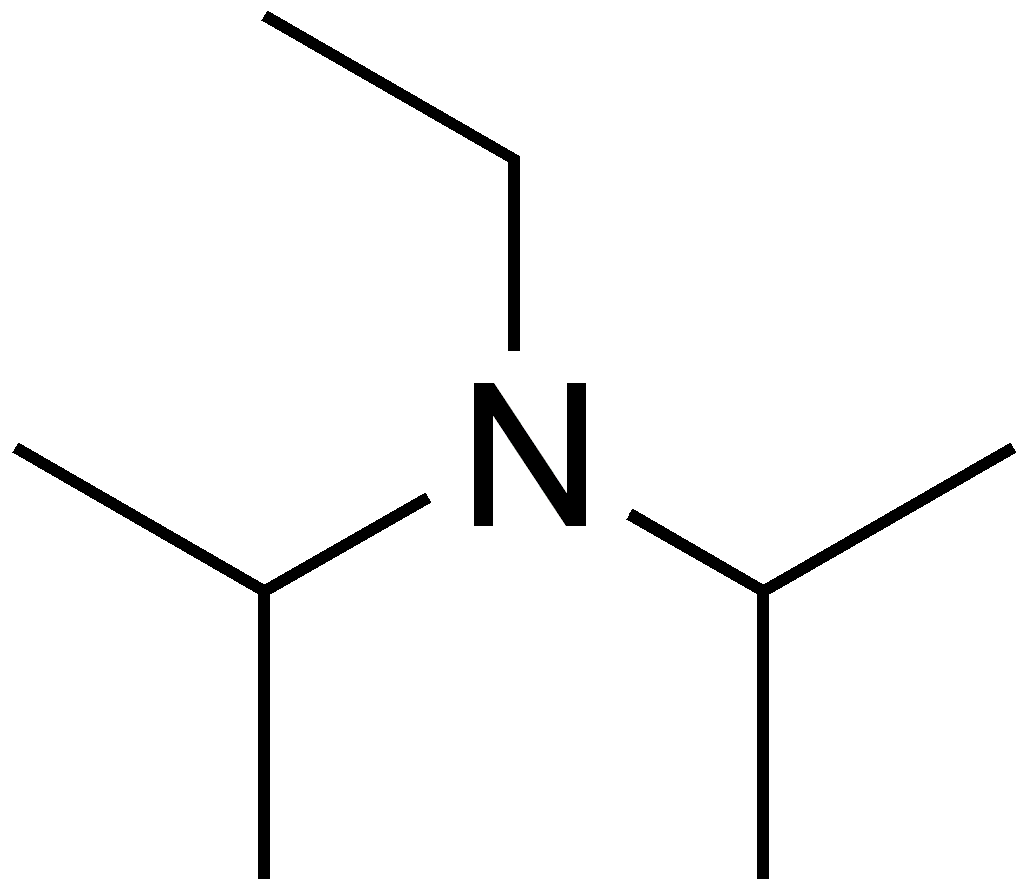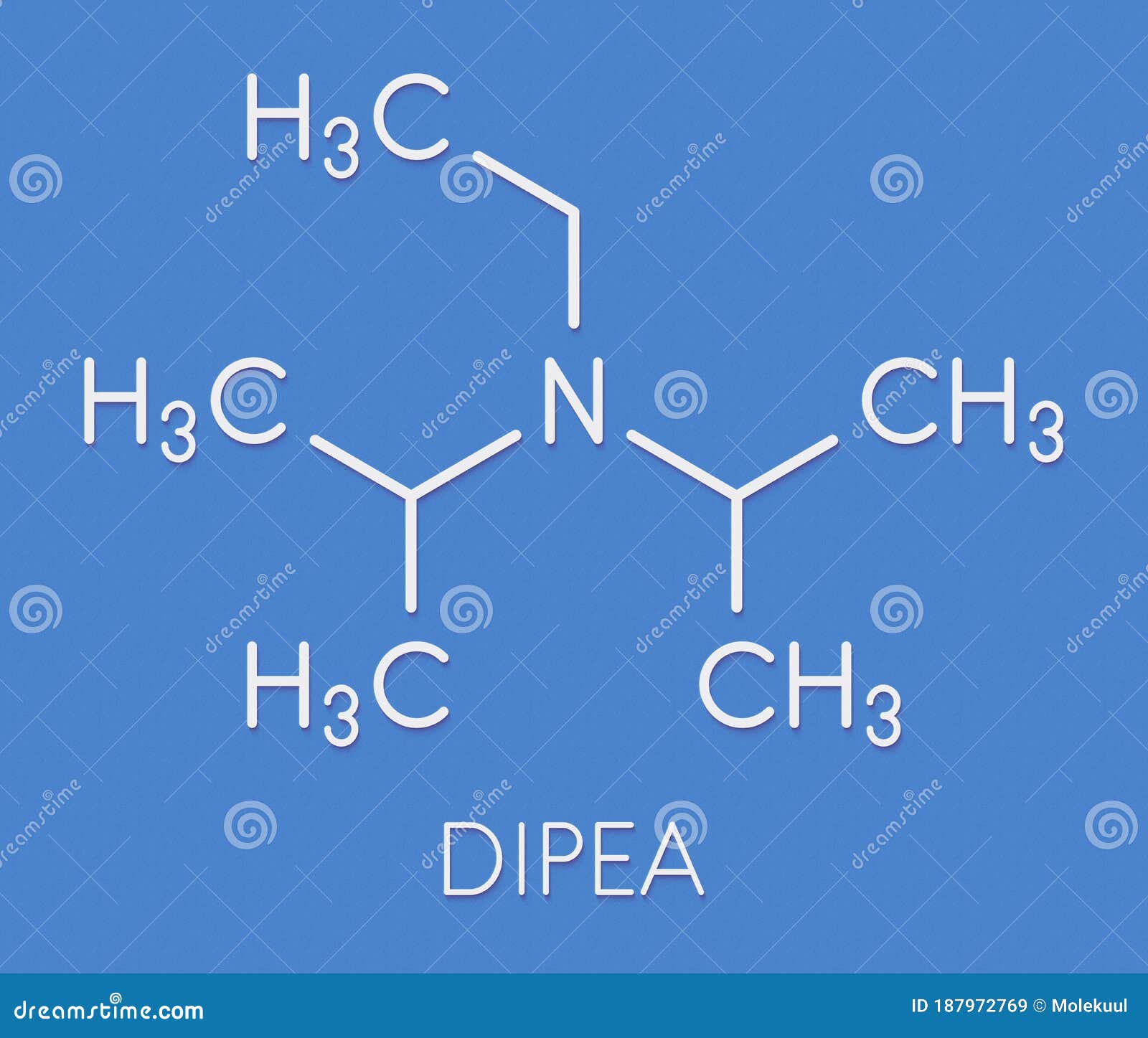
DIPEA N,N-diisopropylethylamine, Hunig`s Base Molecule. Skeletal Formula. Stock Illustration - Illustration of structure, base: 187972769
DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure). Atoms are shown as Stock Photo - Alamy
![PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/37998da1cde9c148c08633c2616b218cf5a07970/4-Table1-1.png)
PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar

DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy

Table 1 from Ru-TsDPEN with formic acid/Hunig's base for asymmetric transfer hydrogenation, a practical synthesis of optically enriched N-propyl pantolactam. | Semantic Scholar

The Story of the Little Blue Box: A Tribute to Siegfried Hünig - Chen - 2023 - Angewandte Chemie International Edition - Wiley Online Library

Dipea Nndiisopropylethylamine Hunigs Base Molecule Skeletal Stock Vector (Royalty Free) 1093026992 | Shutterstock

DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy
Supporting Information Continuous-flow synthesis of primary amines: Metal-free reduction of aliphatic and aromatic nitro derivat

Hunig's base catalyzed synthesis of new 1-(2,3-dihydro-1H-inden-1-yl)-3-aryl urea/thiourea derivatives as potent antioxidants and 2HCK enzyme growth inhibitors - ScienceDirect

Radical Reactions Induced by Visible Light in Dichloromethane Solutions of Hünig's Base: Synthetic Applications and Mechanistic Observations - Böhm - 2016 - Chemistry – A European Journal - Wiley Online Library
![PDF] A SIMPLE AND HIGHLY EFFICIENT SYNTHESIS OF QUINOLINE TERTIARY AMINES CATALYZED BY HUNIG'S BASE | Semantic Scholar PDF] A SIMPLE AND HIGHLY EFFICIENT SYNTHESIS OF QUINOLINE TERTIARY AMINES CATALYZED BY HUNIG'S BASE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3c974f939a32262aeed197ab08f1ab631c012982/4-Table2-1.png)