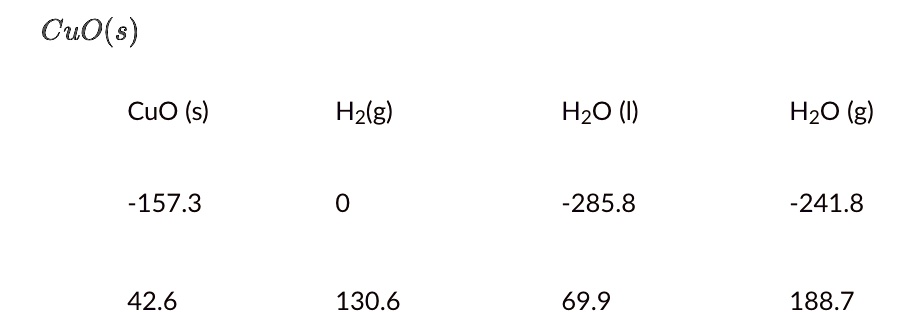
Lab 21Date: Cupric Oxide from Cupric Sulfate Purpose Compare the experimental and calculated amounts of CuO produced in a rxn. Background CuSO 4 (aq) + - ppt download
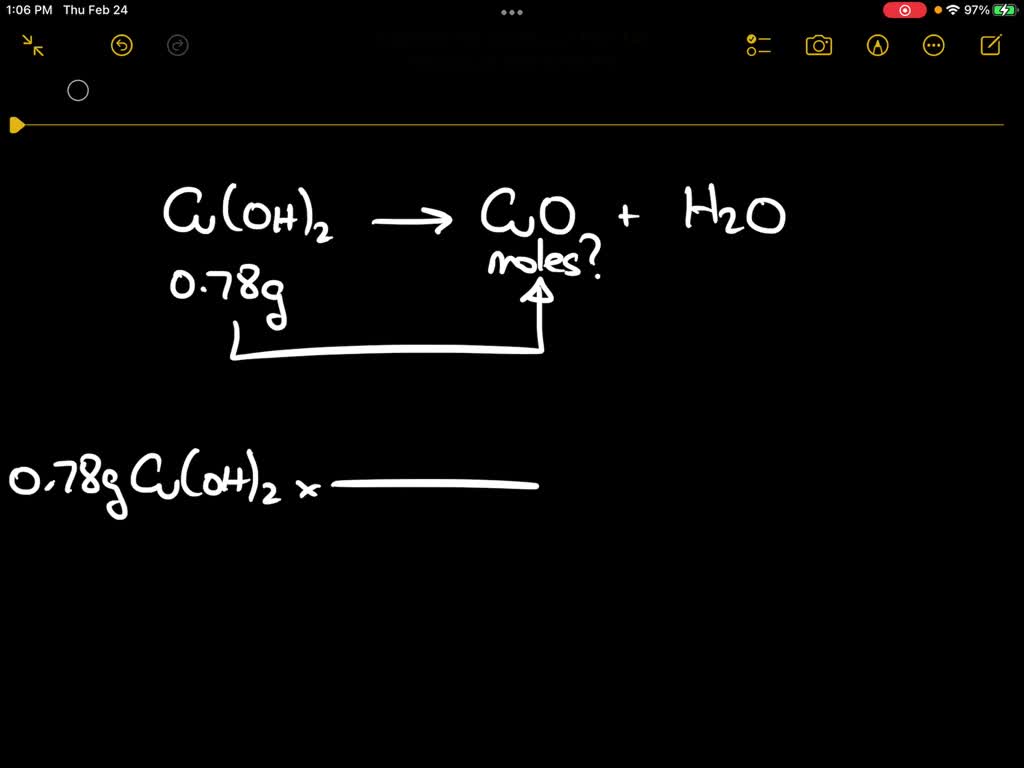
SOLVED: Upon heating Cu(OH)2 decomposes into CuO and H2O as shown below. Cu(OH)2 → CuO + H2O If 0.78 g of Cu(OH)2 was decomposed, calculate the moles of CuO produced Use 29Cu63.5,
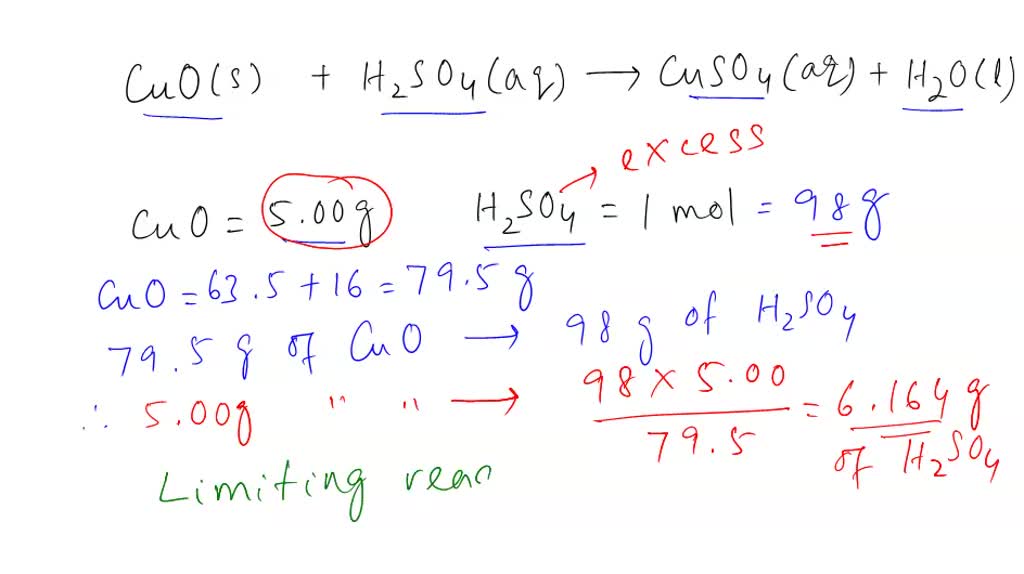
SOLVED: CuO (s) + H2SO4 (aq) → CuSO4 (aq) + H2O (l) When 5.00 g of copper oxide reacts with 1.00 mole of H2SO4, which of the following statements is(are) TRUE? Assume

Pushing the Limits of Rapid Anodic Growth of CuO/Cu(OH)2 Nanoneedles on Cu for the Methanol Oxidation Reaction: Anodization pH Is the Game Changer | ACS Applied Energy Materials