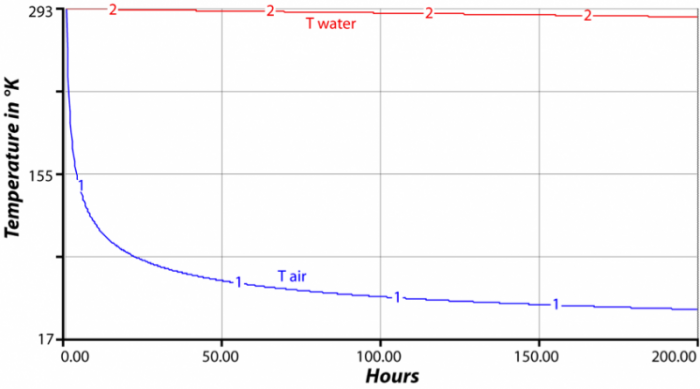
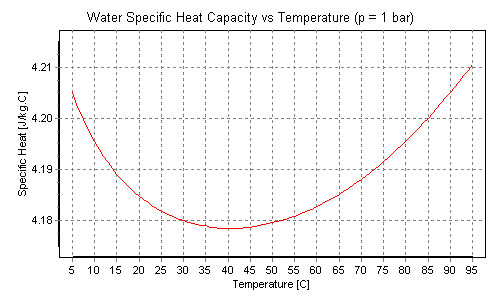
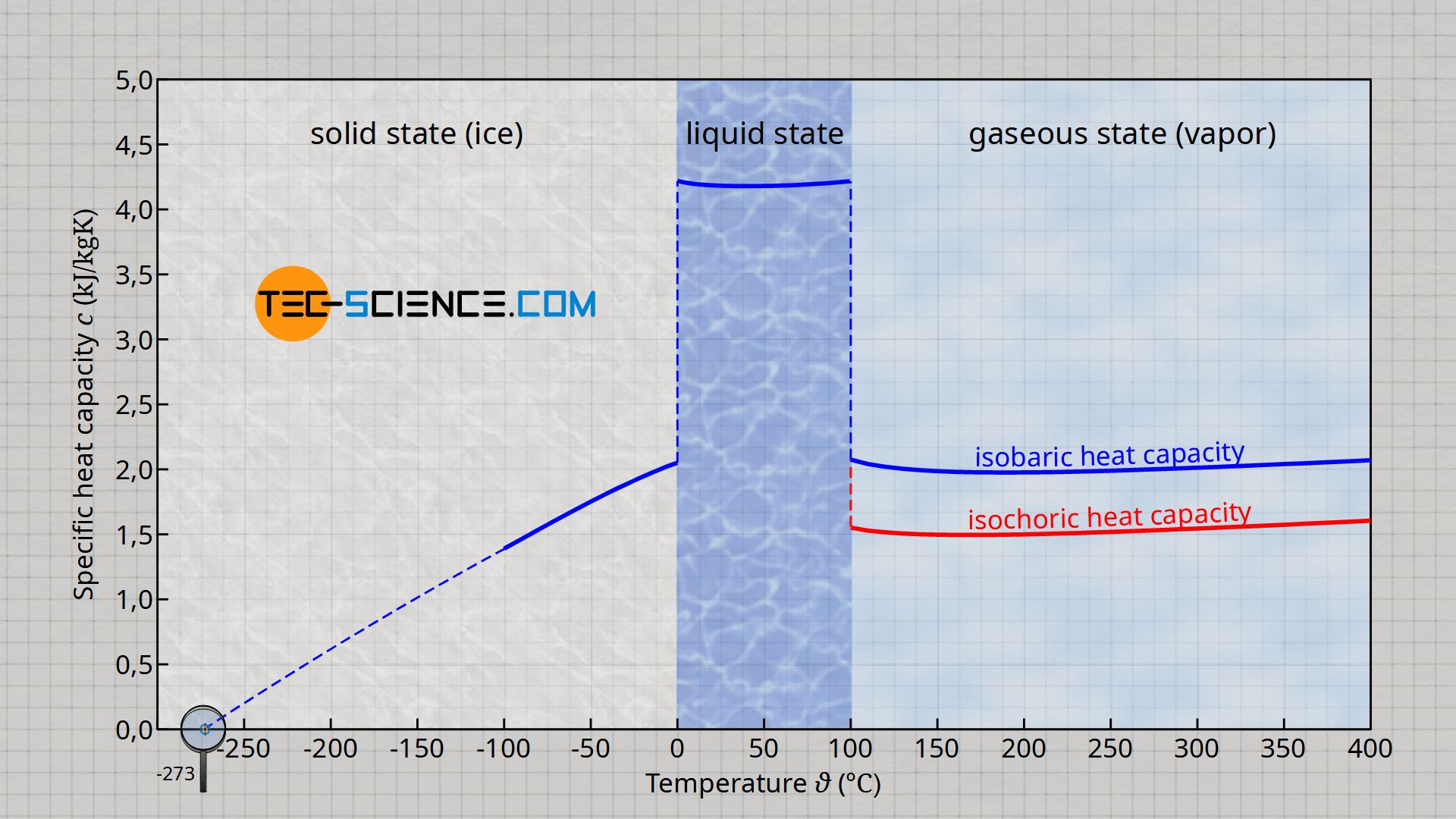
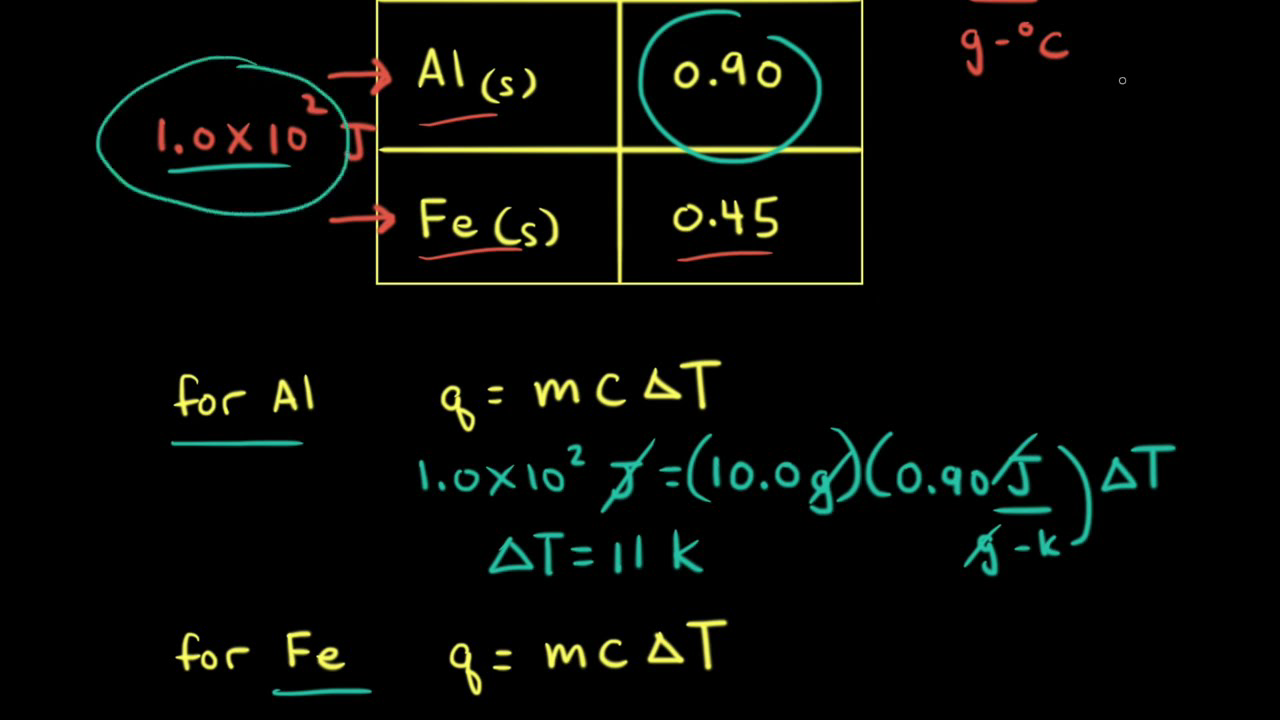SOLVED: Results: Calculate the heat gained by the water (lost by the substance in the calorimeter using the equation in the Introduction: Remember t0 write the unitsl! Q fon water using Ist
![PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e9e458615e33c40d94b80a3189efa26d438c8a39/3-Table1-1.png)
PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar
![Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1) Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1)](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/34965050_web.png)
Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1)

The enthalpy change for a reaction at equilibrium is - 20.5 kJ mol ^-1 . Then the entropy change for this equilibrium at 410 K is:
![Cp*Rh(bpy)(H2O)]2+ as a coenzyme substitute in enzymatic oxidations catalyzed by Baeyer–Villiger monooxygenases - Chemical Communications (RSC Publishing) Cp*Rh(bpy)(H2O)]2+ as a coenzyme substitute in enzymatic oxidations catalyzed by Baeyer–Villiger monooxygenases - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/B504921K)
![Solved Heat Capacities pIH20l 37.65 J/K-mol Cp[H2O(l)]-75.29 | Chegg.com Solved Heat Capacities pIH20l 37.65 J/K-mol Cp[H2O(l)]-75.29 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F9c6%2F9c6322fb-d755-40a1-8987-9484af98164f%2FphpxcNGUz.png)