Synthesis of Spirocyclic Indolenines - James - 2016 - Chemistry – A European Journal - Wiley Online Library

Stereoselective Rearrangements with Chiral Hypervalent Iodine Reagents - Farid - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Coumarin-Caged Compounds of 1-Naphthaleneacetic Acid as Light-Responsive Controlled-Release Plant Root Stimulators | Journal of Agricultural and Food Chemistry

Microstructure evolution and acid corrosion behavior of CoCrFeNiCu1−xMox high-entropy alloy coatings fabricated by coaxial direct laser deposition - ScienceDirect

In Situ Formation of N-Trifluoroacetoxy Succinimide (TFA-NHS): One-Pot Formation of Succinimidyl Esters, N-Trifluoroacetyl Amino Acid Succinimidyl Esters, and N-Maleoyl Amino Acid Succinimidyl Esters | The Journal of Organic Chemistry
Stepwise recombination suppression around the mating-type locus in an ascomycete fungus with self-fertile spores | PLOS Genetics
![Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo400613z/asset/images/jo400613z.social.jpeg_v03)
Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry

In Situ Formation of N-Trifluoroacetoxy Succinimide (TFA-NHS): One-Pot Formation of Succinimidyl Esters, N-Trifluoroacetyl Amino Acid Succinimidyl Esters, and N-Maleoyl Amino Acid Succinimidyl Esters | The Journal of Organic Chemistry
![Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo400613z/asset/images/large/jo-2013-00613z_0010.jpeg)
Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry
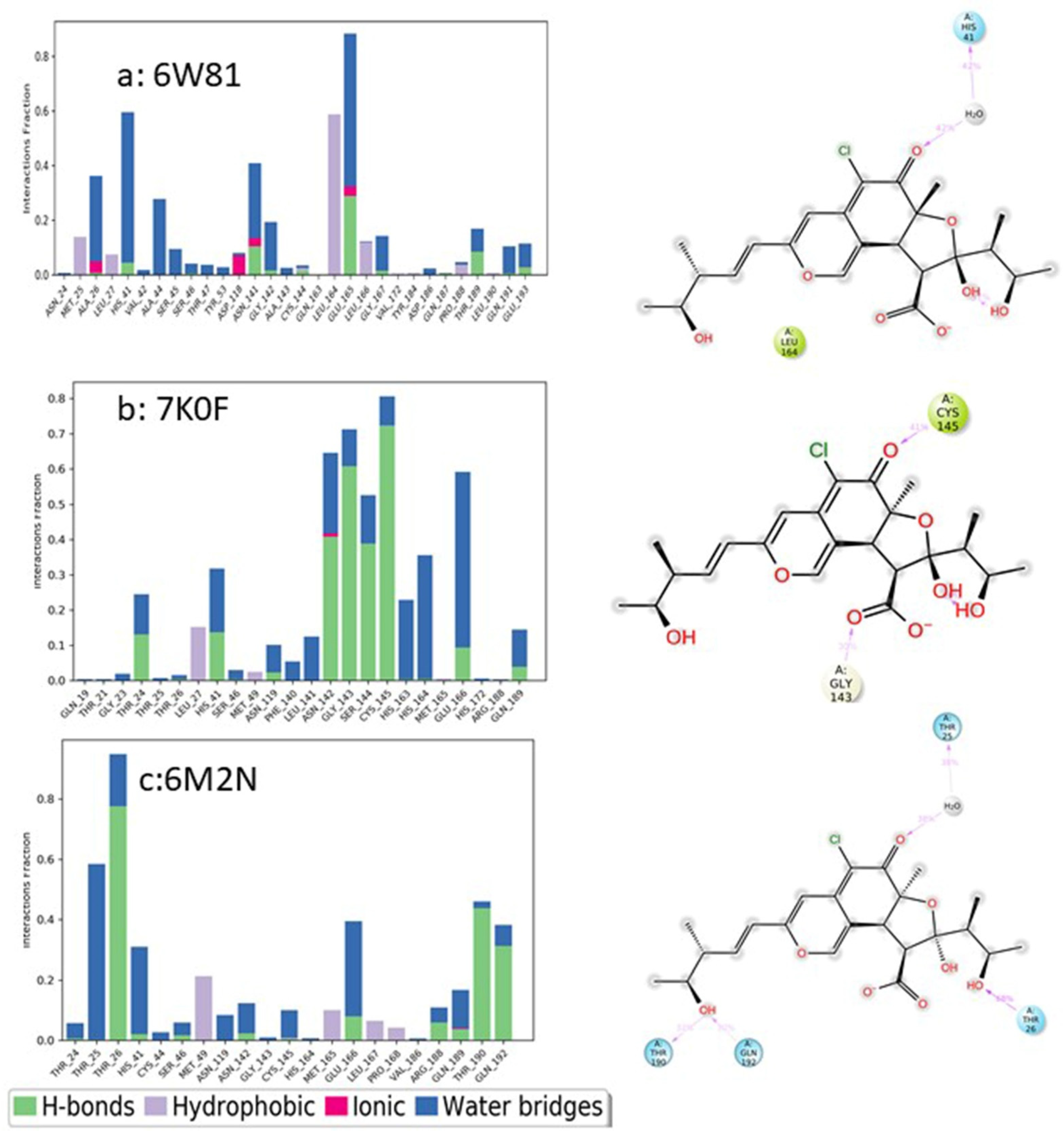
JoF | Free Full-Text | Chaetomugilins and Chaetoviridins—Promising Natural Metabolites: Structures, Separation, Characterization, Biosynthesis, Bioactivities, Molecular Docking, and Molecular Dynamics
![Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo400613z/asset/images/medium/jo-2013-00613z_0010.gif)
Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry
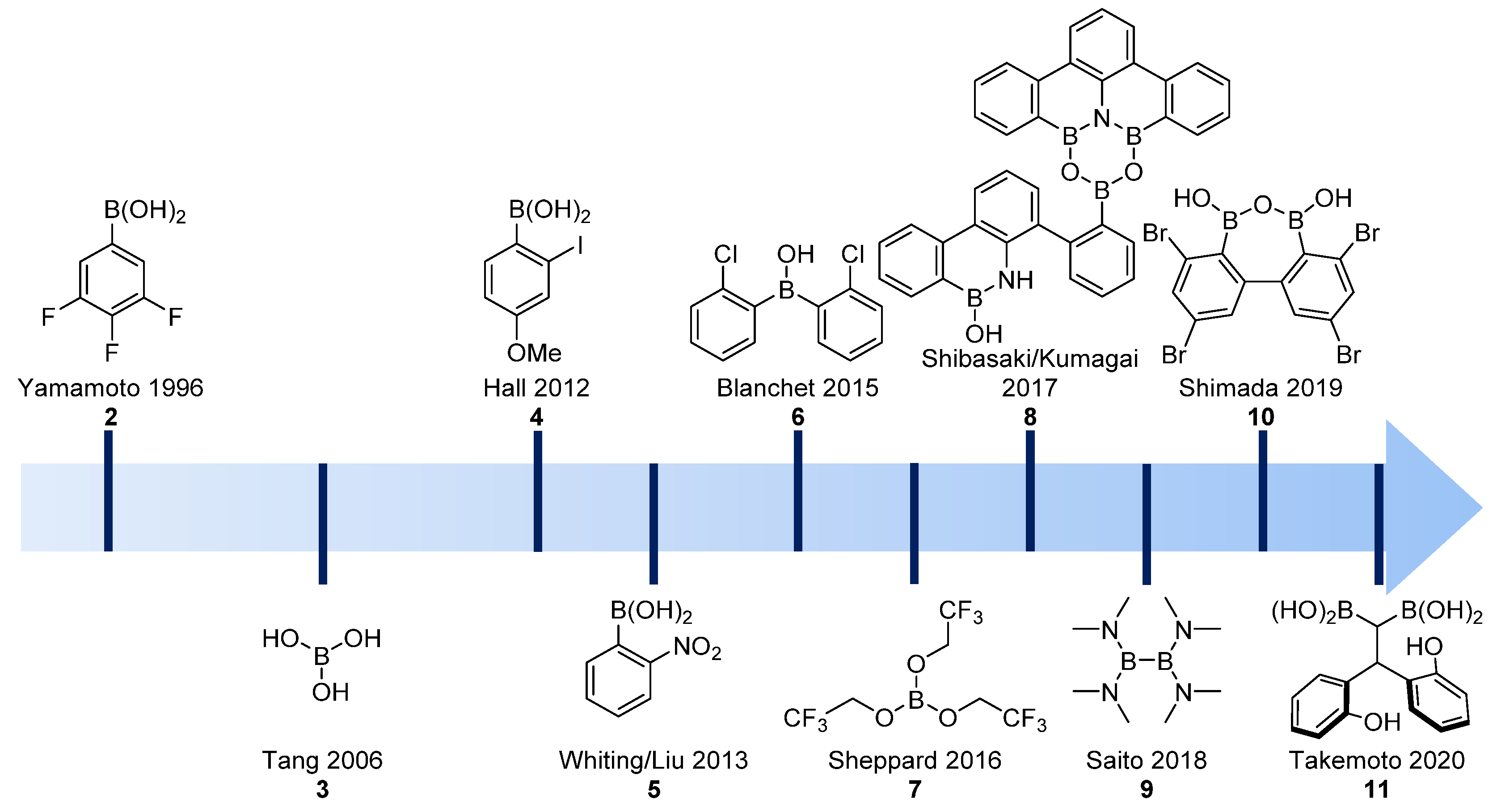
Catalysts | Free Full-Text | Direct Catalytic Amidations from Carboxylic Acid and Ester Derivatives: A Review

Perfluoroaryl and Perfluoroheteroaryl Reagents as Emerging New Tools for Peptide Synthesis, Modification and Bioconjugation - Brittain - 2022 - Chemistry – A European Journal - Wiley Online Library
Perfluoroaryl and Perfluoroheteroaryl Reagents as Emerging New Tools for Peptide Synthesis, Modification and Bioconjugation

Catalysts | Free Full-Text | Direct Catalytic Amidations from Carboxylic Acid and Ester Derivatives: A Review

In Situ Formation of N-Trifluoroacetoxy Succinimide (TFA-NHS): One-Pot Formation of Succinimidyl Esters, N-Trifluoroacetyl Amino Acid Succinimidyl Esters, and N-Maleoyl Amino Acid Succinimidyl Esters | The Journal of Organic Chemistry

Identification of microbes isolated with test kits through culture-dependent and metabarcoding techniques for assessment of microbial corrosion - ScienceDirect
![Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry Brønsted Acid-Controlled [3 + 2] Coupling Reaction of Quinone Monoacetals with Alkene Nucleophiles: A Catalytic System of Perfluorinated Acids and Hydrogen Bond Donor for the Construction of Benzofurans | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo400613z/asset/images/medium/jo-2013-00613z_0020.gif)