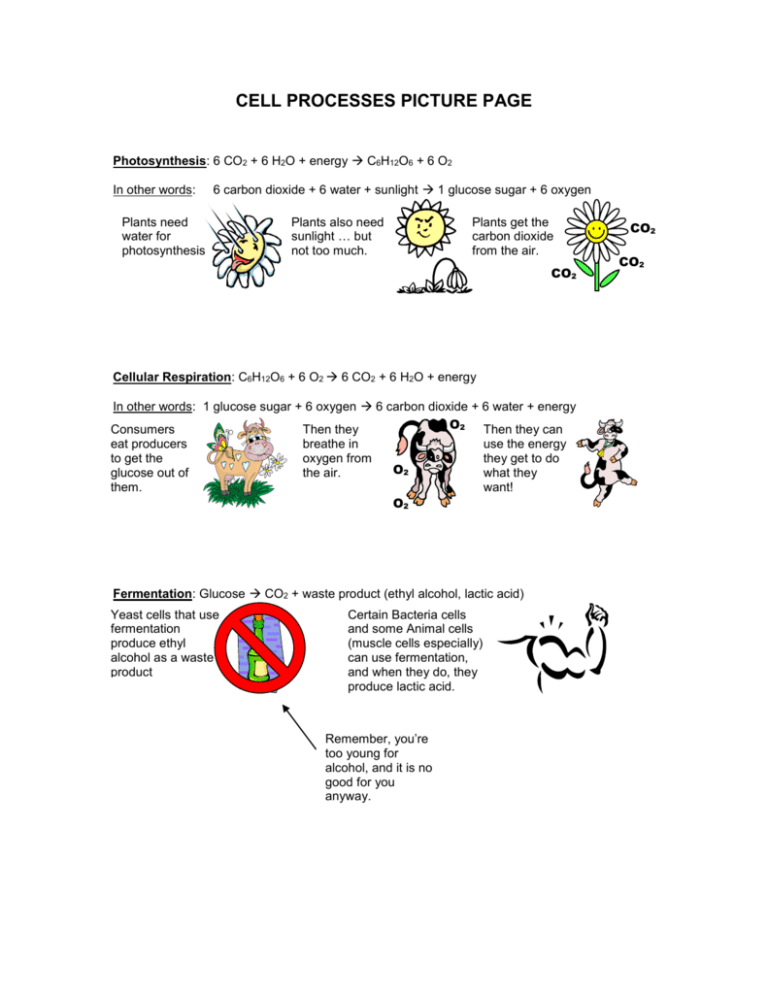
Sodium Phosphate Dibasic Dodecahydrate (Na2HPO4.12H2O) Molecular Weight Calculation - Laboratory Notes

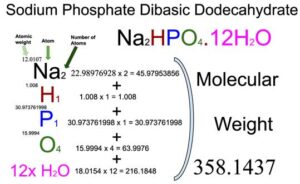
SOLVED: There are several steps to the synthesis of the aluminum potassium alum that can be represented by the overall reaction.2Al(s) + 2KOH(aq) + 4H2SO4(aq) + 24H2O(l) → 2KAl(SO4)212H2O(s) + 3H2(g) +H2O(l)1.

Superpunch Wash Away Stabilizer, SuperStable H2O "Eau" Gone White FSL 12 inch x 10 Yard Roll - Machine Embroidery Stabilizer Backing, Wash n Gone Wash Away for Free Standing Lace, Made in USA

![12" H2O Coil Glass water recycle bong [H2O-18] – Pothead Parent 12" H2O Coil Glass water recycle bong [H2O-18] – Pothead Parent](https://cdn.shopify.com/s/files/1/0605/3917/5125/products/050eed24-12e1-4d34-bb5b-32a8fe97be84.jpg?v=1669868015)
![6CO2 + 12H2O [chlorophyll]sunlight C6H12O6 + 6H2O + 6O2 ↑ The above equation represent 6CO2 + 12H2O [chlorophyll]sunlight C6H12O6 + 6H2O + 6O2 ↑ The above equation represent](https://haygot.s3.amazonaws.com/questions/1469557_451895_ans_7550e023f6324b69a36110ab829282f8.png)